
The rare diseases that
need our attention
We focus on rare diseases where the burden is significant, and the need for better treatment options is clear. By combining scientific insight with a deep understanding of patient experience, we work to advance therapies that can make a meaningful difference in patients’ lives.
Explore our therapeutic areas of focus below.
Rare liver diseases are often complex, progressive, and deeply disruptive to daily life. We have built a leading portfolio in cholestatic liver diseases—addressing a broad range of conditions—while expanding into other rare liver diseases where unmet need remains high. Through our expertise, we develop therapies that target both the underlying disease and the symptoms that matter most to patients.
Select a condition to learn more.
Alagille Syndrome (ALGS)
ALGS is a rare, multisystem disease caused by abnormalities in bile ducts that can lead to progressive liver disease.1
Bile ducts carry bile from the liver to the gallbladder and small intestine.1 Bile has many purposes, including aiding in the digestion of fats and helping with the absorption of fat and certain types of vitamins (A, D, E, and K).2 In people with ALGS, the bile ducts may be too narrow, too few, or missing altogether. This can result in cholestasis: a condition where the flow of bile from the liver is slowed or blocked, causing bile acids to build up in the body.3,4
Over time, the buildup of bile acids in the liver can lead to long-term problems, including inflammation, scar tissue, and injury, as well as the risk for transplant.3,5-7
Approximately 1 in every 30,000 children in the United States and Europe are born with ALGS.8
Signs of ALGS typically begin during infancy and symptoms attributed to cholestasis include2:
- Yellow skin or eyes (jaundice)
- Cholestatic pruritus (itch)
- Stunted growth
Up to 88% of people with ALGS experience cholestatic pruritus (itch).9 Cholestatic pruritus is often a very challenging symptom for people with ALGS, beyond the scratching. It can present in patients differently and range in severity. Signs and symptoms related to the itch can look different during the day and/or night, vary among patients, or even change with age.9,10
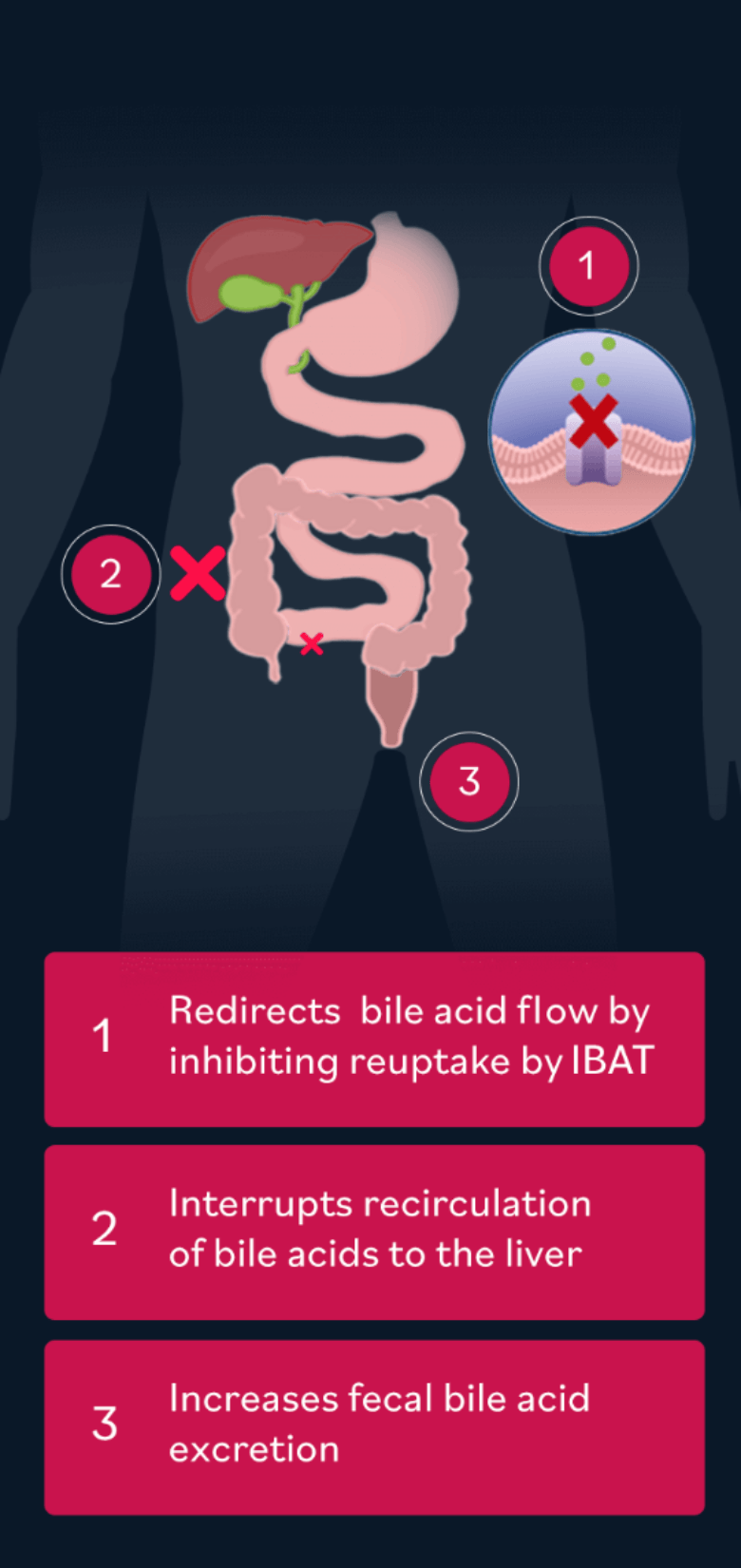
Ileal bile acid transporter (IBAT) inhibition has been approved to treat cholestatic pruritus in patients with ALGS. IBAT inhibition seeks to help redirect bile acids to excrete more in the feces, leading to lower circulating levels and reduced bile buildup in the liver.
References
- Definition & facts for Alagille syndrome. National Institute of Diabetes and Digestive and Kidney Diseases. Updated June 2018. Accessed June 4, 2021. https://www.niddk.nih.gov/health-information/liver-disease/alagille-syndrome/definition-facts
- Alagille syndrome. Johns Hopkins Medicine. Accessed April 29, 2021. https://www.hopkinsmedicine.org/health/conditions-and-diseases/alagille-syndrome
- Kamath BM, Stein P, Houwen RHJ, Verkade HJ. Potential of ileal bile acid transporter inhibition as a therapeutic target in Alagille syndrome and progressive familial intrahepatic cholestasis. Liver Int. 2020;40(8):1812-1822. doi:10.1111/liv.14553
- Alagille syndrome. MedlinePlus. Updated December 1, 2023. Accessed February 7, 2024. https://medlineplus.gov/genetics/condition/alagille-syndrome
- Cai SY. The role of bile acids in cholestatic liver injury. Ann Transl Med. 2021;9:737. doi:10.21037/atm-20-5110
- Karpen SJ, Kelly D, Mack C, Stein P. Ileal bile acid transporter inhibition as an anticholestatic therapeutic target in biliary atresia and other cholestatic disorders. Hepatol Int. 2020;14(5):677-689. doi:10.1007/s12072-020-10070-w
- Li T, Chiang J. Bile acid-induced liver injury in cholestasis. In: Ding WX, Yin XM, eds. Cellular Injury in Liver Diseases. Cell Death in Biology and Diseases. Springer; 2017:143-172. doi:10.1007/978-3-319-53774-0_7
- Vandriel SM, Li LT, She H, Wang JS, et al; Global Alagille Alliance (GALA) Study Group. Natural history of liver disease in a large international cohort of children with Alagille syndrome: results from the GALA study. Hepatology. 2023;77(2):512-529. doi:10.1002/hep.32761
- Kamath BM, Baker A, Houwen R, Todorova L, Kerkar N. Systematic review: the epidemiology, natural history, and burden of Alagille syndrome. J Pediatr Gastroenterol Nutr. 2018;67(2):148-156. doi:10.1097/MPG.0000000000001958
- Langedijk JAGM, Beuers UH, Oude Elferink RPJ. Cholestasis-associated pruritus and its pruritogens. Front Med (Lausanne). 2021;8:639674. doi:10.3389/fmed.2021.639674
Progressive familial intrahepatic cholestasis (PFIC)
PFIC is a group of rare, inherited, genetic diseases that mostly affect the liver. PFIC is characterized by impaired bile acid secretion or transport, leading to progressive cholestatic liver disease. Over time, the buildup of bile acids in the liver can lead to long-term problems, including inflammation, scar tissue, and injury, as well as the risk for transplant.1
PFIC is classified into three main subtypes along with several rare subtypes. These subtypes are defined by underlying genetic mutations, such as in the ABCB11 or ABCB4 genes, which, when altered, cause bile acids to accumulate in the liver.2,3 Across all PFIC subtypes, there is an inhibition of bile flow between the liver and small intestine, resulting in a persistent state of cholestasis.4,5
Signs of PFIC can begin as early as infancy and can result in:
- Cholestatic pruritus (itch)2
- Stunted growth1,2
- Vitamin deficiency1
- Yellow skin or eyes (jaundice)2
- Progressive liver damage2
- Liver failure if left untreated2
Though PFIC typically appears in infancy or early childhood, with some types, like PFIC3, symptoms may not present until adulthood. PFIC can also be missed in adults, and symptoms like cholestatic pruritus are often normalized or overlooked, adding to diagnosis challenges.2,6
Up to 100% of people with PFIC are affected by cholestatic pruritus and it is also the most burdensome symptom.7 Uncontrolled cholestatic pruritus causes more than just skin damage. Patients can also struggle with fatigue, irritability, physical discomfort, decreased physical function, impaired school performance, and negative impact on social activities.1,7
There are fewer than 1,000 pediatric patients with PFIC in either the United States or Europe.8
Ileal bile acid transporter (IBAT) inhibition has been approved to treat cholestatic pruritus in patients with PFIC.* IBAT inhibition seeks to help redirect bile acids to excrete more in the feces, leading to lower circulating levels and reduced bile buildup in the liver.

*IBAT is not recommended in a subgroup of PFIC patients with non-functional or complete absence of BSEP protein.
References
- Srivastava A. Progressive familial intrahepatic cholestasis. J Clin Exp Hepatol. 2014;4(1):25-36. doi:10.1016/j.jceh.2013.10.005
- Davit-Spraul A, Gonzales E, Baussan C, Jacquemin E. Progressive familial intrahepatic cholestasis. Orphanet J Rare Dis. 2009;4:1. doi:10.1186/1750-1172-4-1
- Vinayagamoorthy V, Srivastava A, Sarma MS. Newer variants of progressive familial intrahepatic cholestasis. World J Hepatol. 2021;13(12):2024-2038. doi:10.4254/wjh.v13.i12.2024
- Felzen A, Verkade HJ. The spectrum of progressive familial intrahepatic cholestasis diseases: update on pathophysiology and emerging treatments. Eur J Med Genet. 2021;64(11):104317. doi:10.1016/j.ejmg.2021.104317
- Kamath BM, Stein P, Houwen RHJ, Verkade HJ. Potential of ileal bile acid transporter inhibition as a therapeutic target in Alagille syndrome and progressive familial intrahepatic cholestasis. Liver Int. 2020;40(8):1812-1822. doi:10.1111/liv.14553
- PFIC diagnosis – types of tests. PFIC Network. Accessed March 30, 2026. https://www.pfic.org/learn-about-pfic-disease/pfic-diagnosis/
- Baker A, Kerkar N, Todorova L, Kamath BM, Houwen RHJ. Systematic review of progressive familial intrahepatic cholestasis. Clin Res Hepatol Gastroenterol. 2019;43(1):20-36. doi:10.1016/j.clinre.2018.07.010c
- Data on File. Mirum Pharmaceuticals.
Primary sclerosing cholangitis (PSC)
PSC is a rare cholestatic liver disease characterized by inflammation and scarring of the bile ducts, which lead to hardening and narrowing (strictures) in the bile ducts inside or outside the liver. This prevents the bile from flowing properly, ultimately requiring a liver transplant for many patients.1 These blockages can cause bile acids to build up in the body—a condition known as cholestasis. This leads to higher levels of bile acids in the blood, which are linked to common symptoms like itching and fatigue.2
PSC affects approximately 10 in 100,000 people, and nearly 70% of those patients also have inflammatory bowel disease.3,4
Signs and symptoms of PSC change as the disease progresses and may include:
EARLY SYMPTOMS
- Itch (pruritus)5
- Fatigue5
- Brain fog6
- Upper right quadrant abdominal pain5
- Episodes of cholangitis (an infection within the liver)7
- Chills and fever5
LATER SYMPTOMS
- Ascites (swollen abdomen with fluid)8
- Enlarged spleen (splenomegaly)4
- Yellowing of skin and eyes (jaundice)8
- Unintended weight loss4
- Liver failure5
Up to 91% of PSC patients experience itch at some point during the course of their disease, often linked to sleep disturbance, fatigue, and mood changes.9 Many PSC patients will experience pruritus (itching) and it tends to worsen as the disease progresses. While itch is a common symptom of PSC, it is often underreported.10,11
Despite the impact of PSC on a patient’s quality of life, there are currently no approved treatments that can slow or stop PSC from progressing or treat the persistent itch and fatigue that typically accompany the disease.8 To address this unmet need, we are investigating a selective inhibitor of the ileal bile acid transporter (IBAT) to treat cholestasis. IBAT inhibition seeks to help redirect bile acids to excrete more in the feces, leading to lower circulating levels and reduced bile buildup in the liver.

References
- Mayo Foundation for Medical Education and Research. Primary sclerosing cholangitis: Symptoms and causes. Mayo Clinic. Published June 21, 2023. Accessed June 2, 2025. https://www.mayoclinic.org/diseases-conditions/primary-sclerosing-cholangitis/symptoms-causes/syc-20355797
- Sanjel B, Shim WS. Recent advances in understanding the molecular mechanisms of cholestatic pruritus: a review. Biochim Biophys Acta Mol Basis Dis. 2020;1866(12):165958. doi:10.1016/j.bbadis.2020.165958
- Sohal A, Kayani S, Kowdley KV. Primary sclerosing cholangitis: epidemiology, diagnosis, and presentation. Clin Liver Dis. 2024;28(1):129-141. doi:10.1016/j.cld.2023.07.005
- Primary sclerosing cholangitis: MedlinePlus Genetics. Accessed April 1, 2026. https://medlineplus.gov/genetics/condition/primary-sclerosing-cholangitis/
- What is primary sclerosing cholangitis? Cleveland Clinic. Accessed March 31, 2026. https://my.clevelandclinic.org/health/diseases/23569-primary-sclerosing-cholangitis
- Arndtz K, Cameron M, Hirschfield G, Parry J, Greenfield S. What are the lived healthcare experiences of patients with primary sclerosing cholangitis? A community-based qualitative interview study. BMJ Open. 2025;15(2):e082498. doi:10.1136/bmjopen-2023-082498
- Chazouilleres O, Beuers U, Bergquist A, et al. EASL Clinical Practice Guidelines on sclerosing cholangitis. J Hepatol. 2022;77(3):761-806. doi:10.1016/j.jhep.2022.05.011
- Lindor KD, Kowdley KV, Harrison ME; American College of Gastroenterology. ACG clinical guideline: primary sclerosing cholangitis. Am J Gastroenterol. 2015;110(5):646-659. doi:10.1038/ ajg.2015.112
- Kowdley KV, Safer R, Gomel R, et al. Impact of pruritus in primary sclerosing cholangitis (PSC): a multinational survey. Poster presented at: The International Liver Congress; 2022. Accessed June 2, 2025 https://www.postersessiononline.eu/173580348_eu/congresos/ILC2022/aula/-THU_442_ILC2022.pdf
- Dean R, Yazdanfar M, Zepeda J, et al. Treatment of pruritus in primary sclerosing cholangitis: analysis of the consortium for autoimmune liver disease registry. Hepatol Commun. 2025;9(5):e0703. doi:10.1097/HC9.0000000000000703
- Hussain N, Motta RV, Gungabissoon U, et al. Pruritus is common in primary sclerosing cholangitis, persists over time, and its intensity is associated with disease severity: a multicentre, prospective observational study. Hepatol. Published online December 5, 2025.
Primary biliary cholangitis (PBC)
PBC is a rare cholestatic liver disease that causes the small bile ducts in the liver to become injured and inflamed. When bile ducts become injured, bile builds up (cholestasis) and potentially causes progressive liver damage, with the need for liver transplant in some patients.1 Cholestasis is linked to common symptoms such as itch and fatigue.2
PBC is thought to be an autoimmune disease in which the body attacks its own bile ducts. People with a family member who has PBC may also have a higher chance of developing the disease.1
Early and general symptoms include3:
- Fatigue
- Itch (pruritus)
- Abdominal pain
Later signs and symptoms of PBC include3:
- Jaundice
- Enlarged spleen (splenomegaly)
- Swelling in the feet and ankles
People living with PBC typically rate the itch and fatigue they experience as moderate to severe.4,5 The burdensome itch can significantly impact a patient’s quality of life, affecting their social and emotional well-being and contributing to sleep disruption that worsens fatigue.5 While itch and fatigue are common symptoms of PBC, they are often underreported.4,5
There is no cure for PBC, but medications may help manage the disease and slow the progression of liver damage. Other medications are typically prescribed to help manage a person’s symptoms, including itch. When medications do not help, liver transplantation may be necessary.6 Mirum is investigating a selective inhibitor of the ileal bile acid transporter (IBAT) to treat cholestasis. IBAT inhibition seeks to help redirect bile acids to excrete more in the feces, leading to lower circulating levels and reduced bile buildup in the liver.

References
- Mayo Foundation for Medical Education and Research. Primary biliary cholangitis. Mayo Clinic. Published November 14, 2023. Accessed June 2, 2025. https://www.mayoclinic.org/diseases-conditions/primary-biliary-cholangitis/symptoms-causes/syc-20376874
- Faisal A. Understanding fatigue and pruritus in primary biliary cholangitis. Clin Liver Dis (Hoboken). 2024;23(1):e0216. doi:10.1097/CLD.0000000000000216
- Lindor KD, Bowlus CL, Boyer J, Levy C, Mayo M. Primary Biliary Cholangitis: 2018 Practice Guidance from the American Association for the Study of Liver Diseases. Hepatology. 2019;69(1):394-419. doi:10.1002/hep.30145
- Al-Harthy N, Kumagi T, Coltescu C, Hirschfield GM. The specificity of fatigue in primary biliary cirrhosis: evaluation of a large clinic practice. Hepatology. 2010;52(2):562-570. doi:10.1002/hep.23683
- Gungabissoon U, Smith HT, von Maltzahn R, et al. Pruritus in primary biliary cholangitis is under-recorded in patient medical records. BMJ Open Gastroenterol. 2024;11(1):e001287. doi:10.1136/bmjgast-2023-001287
- National Organization for Rare Disorders. Primary biliary cholangitis. NORD. Updated April 6, 2020. Accessed June 1, 2025. https://rarediseases.org/rare-diseases/primary-biliary-cholangitis/
Pruritus in ultra-rare cholestatic settings
Cholestatic pruritus is a debilitating symptom associated with a range of rare liver diseases in which bile flow is reduced or blocked, leading to the buildup of bile acids in the body (cholestasis).1,2 The resulting itch is often persistent and difficult to manage.3
Patients living with cholestatic pruritus may experience:
- Unrelenting itching that can feel deep beneath the skin3,4
- Sleep disruption3
- Chronic fatigue1
- Difficulty concentrating and reduced daily functioning5
- Emotional and social impacts on quality of life3,5
Despite its impact, treatment options have historically been limited. Targeting the ileal bile acid transporter (IBAT) represents a promising approach to reducing bile acid levels and addressing the underlying driver of cholestatic pruritus, an approach that has been associated with improvements in itch, which affects patients’ quality of life.3,6
References
- Hilscher MB, Kamath PS, Eaton JE. Cholestatic liver diseases: a primer for generalists and subspecialists. Mayo Clin Proc. 2020;95(10):2263-2279. doi:10.1016/j.mayocp.2020.01.015
- Cholestasis: definition, symptoms, treatment, causes. Cleveland Clinic. Accessed April 1, 2026. https://my.clevelandclinic.org/health/diseases/24554-cholestasis
- Patel SP, Vasavda C, Ho B, Meixiong J, Dong X, Kwatra SG. Cholestatic pruritus: emerging mechanisms and therapeutics. J Am Acad Dermatol. 2019;81(6):1371-1378. doi:10.1016/j.jaad.2019.04.035
- Hegade VS, Boiler R, Oude Elferink RP, Beuers U, Kendrick S, Jones DE. a systematic approach to the management of cholestatic pruritus in primary biliary cirrhosis. Frontline Gastroenterol. 2016;7(3):158-166. doi:10.1136/flgastro-2015-100618
- Jin XY, Khan TM. Quality of life among patients suffering from cholestatic liver disease-induced pruritus: A systematic review. J Formos Med Assoc. 2016;115(9):689-702. doi:10.1016/j.jfma.2016.05.006
- Loomes KM, Squires RH, Kelly D, et al. Maralixibat for the treatment of PFIC: long‐term, IBAT inhibition in an open‐label, phase 2 study. Hepatol Commun. 2022;6(9):2379-2390. doi:10.1002/hep4.1980
Hepatitis delta virus (HDV)
HDV, an infection that occurs in some people infected with the hepatitis B virus, is the most severe form of viral hepatitis due to the potential for rapid progression to liver cirrhosis, liver cancer, and liver-related death.1
HDV affects approximately 230,000 people in the United States and Europe.2
HDV often accelerates liver damage beyond that caused by hepatitis B alone.3 People with HDV may experience persistent fatigue, abdominal discomfort, jaundice (yellowing of the skin or eyes), and unexplained weight loss.3-5
Over time, ongoing inflammation can lead to progressive liver scarring (fibrosis), cirrhosis, and increased risk of liver failure or liver cancer, with symptoms and severity varying from person to person.6
There are currently no approved treatments for HDV in the United States and most countries worldwide.
References
- Stockdale AJ, Kreuels B, Henrion MYR, et al. The global prevalence of hepatitis D virus infection: systematic review and meta-analysis. J Hepatol. 2020;73(3):523-532. doi:10.1016/j.jhep.2020.04.008
- Data on File. Mirum Pharmaceuticals.
- Miao Z, Zhang S, Ou X, et al. Estimating the global prevalence, disease progression, and clinical outcome of hepatitis delta virus infection. J Infect Dis. 2020;221(10):1677-1687. doi:10.1093/infdis/jiz633
- Olsen K, Mahgoub S, Al-Shakhshir S, et al. Recent treatment advances and practical management of hepatitis D virus. Clin Med. 2023;23(4):403-408. doi:10.7861/clinmed.2022-0556
- Hepatitis D: Symptoms, diagnosis and treatment | BMJ Best Practice. https://bestpractice.bmj.com/topics/en-gb/3000320
- Negro F, Lok AS. Hepatitis D: a review. JAMA. 2023;330(24):2376-2387. doi:10.1001/jama.2023.23242
Rare genetic diseases are often lifelong, affecting multiple systems and placing a significant burden on patients and families. We are focused on conditions driven by well-defined genetic and metabolic pathways, advancing treatments that target underlying disease biology with the goal of delivering meaningful impact for communities that have long been overlooked and underserved.
Select a condition to learn more.
Bile acid synthesis disorders (BASDs)
BASDs are a group of rare, inherited metabolic disorders caused by defects in the enzymes that are involved in making bile acids. Bile acids promote the flow and excretion of bile, excess cholesterol and waste, and help the intestines absorb fat and fat-soluble vitamins.1
When the body is unable to produce normal bile acids, it can result in interrupted bile flow (cholestasis), often resulting in malabsorption of vital nutrients and the buildup of toxic materials in the body. BASDs are typically detected during infancy and if left untreated, buildup of toxic materials may result in progressive liver disease.1
Cholestasis may be accompanied by the following signs and symptoms1:
- Yellow skin or eyes (jaundice)
- Failure to thrive
- Growth deficiency
- Diarrhea and pale, clay-colored stools caused by lack of bile
- Weakened bones caused by vitamin D deficiency
- Easy bleeding and bruising due to blood clotting issues from vitamin K deficiency
- Vision problems due to vitamin A deficiency
Reference
- National Organization for Rare Disorders. Bile acid synthesis disorders. NORD. Published January 12, 2023. Accessed June 1, 2025. https://rarediseases.org/rare-diseases/bile-acid-synthesis-disorders
Peroxisomal biogenesis disorder-zellweger spectrum disorder
(PBD-ZSD)
PBD-ZSD is a rare, genetic condition caused by the loss of peroxisome function.1 Peroxisomes help produce bile acids, which are important to help the liver function correctly and eliminate waste from the body.2
In PBD-ZSD, nonworking peroxisomes cannot produce healthy bile acids, causing toxic bile acids to get trapped in the liver. This buildup stops or slows the flow of bile from the liver, a condition called cholestasis, which can lead to inflammation, liver damage, and cirrhosis (scarring of the liver).3
PBD-ZSD occurs in an estimated 1 in 50,000 to 75,000 births in the United States.4
Signs and symptoms of liver involvement include:
- Yellow skin or white part of the eyes (jaundice)5
- Not meeting weight or height milestones for age (poor growth)1
- Fat-soluble vitamin deficiencies (vitamins A, D, E, or K)1
- Pale, foul-smelling stools1,6
- Dark urine (tea-colored)1,6
- Enlarged liver or spleen7
- Elevated liver enzymes5
- Bile flow impairment (cholestasis)5
Bile acid replacement therapy could help improve the level of bile acids in the body, which helps the liver absorb vitamins and nutrients that the body needs.1
References
- Braverman NE, Raymond GV, Rizzo WB, et al. Peroxisome biogenesis disorders in the Zellweger spectrum: an overview of current diagnosis, clinical manifestations, and treatment guidelines. Mol Genet Metab. 2016;117(3):313-321. doi:10.1016/j.ymgme.2015.12.009
- Klouwer FC, Berendse K, Ferdinandusse S, Wanders RJ, Engelen M, Poll-The BT. Zellweger spectrum disorders: clinical overview and management approach. Orphanet J Rare Dis. 2015;10:151. doi:10.1186/s13023-015-0368-9
- Ferdinandusse S, Denis S, Faust PL, Wanders RJ. Bile acids: the role of peroxisomes. J Lipid Res. 2009;50(11):2139-2147. doi:10.1194/jlr.R900009-JLR200
- National Organization for Rare Disorders. Zellweger spectrum disorders. NORD. Updated August 18, 2020. Accessed June 9, 2025. https://rarediseases.org/rare-diseases/zellweger- spectrum-disorders/
- Enns GM, Ammous Z, Himes RW, et al. Diagnostic challenges and disease management in patients with a mild Zellweger spectrum disorder phenotype. Mol Genet Metab. 2021;134(3):217-222. doi:10.1016/j.ymgme.2021.09.007
- Tholey D. Cholestasis. Merck Manual Consumer Version. Revised January 2023. Accessed June 9, 2025. https://www.merckmanuals.com/home/liver-and-gallbladder-disorders/ manifestations-of-liver-disease/cholestasis
- MedlinePlus. Bethesda (MD): U.S. National Library of Medicine. Zellweger spectrum disorder. Accessed June 9, 2025. https://medlineplus.gov/genetics/condition/zellweger-spectrum-disorder
Smith-lemli-opitz syndrome (SLOS)
SLOS is a rare genetic condition caused by a mutation in the 7-dehydrocholesterol reductase (DHCR7) gene that helps the body to produce cholesterol. Patients with SLOS are unable to make sufficient amounts of cholesterol, an essential nutrient for proper growth and development.1
Because cholesterol is also necessary for the production of bile acids in the liver, it is likely that low cholesterol levels in SLOS impair bile acids from being made. Bile acids help digest dietary cholesterol from the intestine. Low bile acid levels may prevent dietary cholesterol from being absorbed properly and contribute to the cholesterol deficiency seen in SLOS.1
In addition to low cholesterol absorption that can delay growth and development, some patients may experience elevated or abnormal liver function levels, such as aspartate aminotransferase (AST) and alanine transaminase (ALT), which are important measurements of liver health.1
SLOS affects about 1 in 20,000 to 1 in 60,000 live births.2-6
References
- Nowaczyk MJ, Wassif CA. Smith-lemli-opitz syndrome. In: Adam MP, Feldman J, Mirzaa GM, Pagon RA, Wallace SE, Amemiya A, eds. GeneReviews®. University of Washington, Seattle; 1993. Accessed June 20, 2025. http://www.ncbi.nlm.nih.gov/books/NBK1143/
- Waterham HR, Hennekam RC. Mutational spectrum of Smith-Lemli-Opitz syndrome. Am J Med Genet C Semin Med Genet. 2012;160C(4):263-284. doi:10.1002/ajmg.c.31346
- Opitz JM. RSH (so-called Smith-Lemli-Opitz) syndrome. Curr Opin Pediatr. 1999;11(4):353-362. doi:10.1097/00008480-199908000-00015.
- Nowaczyk MJ, McCaughey D, Whelan DT, Porter FD. Incidence of Smith-Lemli-Opitz syndrome in Ontario, Canada. Am J Med Genet. 2001;102(1):18-20. doi:10.1002/1096-8628(20010722)102:1<18::aid-ajmg1376>3.0.co;2-e
- National Organization for Rare Disorders. Smith-Lemli-Opitz syndrome. NORD. Updated November 17, 2021. Accessed August 8, 2025. https://rarediseases.org/rare-diseases/smith-lemliopitz-syndrome/#affected
- Kritzer A, Dutta R, Pramparo T, Terner-Rosenthal J, Vig P, Steiner RD. Smith-Lemli-Opitz syndrome: clinical, biochemical, and genetic insights with emerging treatment opportunities. Genet Med. 2025;27(7):101450. doi:10.1016/j.gim.2025.101450
Cerebrotendinous xanthomatosis (CTX)
CTX is a rare, genetic, lipid storage and metabolic disorder affecting many parts of the body. CTX stems from a mutated CYP27A1 gene, which causes a deficiency in an enzyme (sterol 27-hydroxylase) needed for bile acid synthesis—specifically to replenish the chenodeoxycholic acid (CDCA) levels that CTX depletes.1,2
Without sufficient CDCA, negative feedback mechanisms that regulate bile acid synthesis are inhibited. This causes an overproduction of toxic bile acid precursors (e.g., 23S-pentol, cholestanol).2,3
Signs and symptoms of CTX include4:
- Neonatal cholestasis (jaundice or bile flow interruption)
- Frequent diarrhea
- Development of cataracts in both eyes before the age of 18
- Development of tendon xanthomas (fatty deposits in the tendons) during teenage years or later
- Neurological problems
The types, combinations, and severity of symptoms can be different from person-to-person, making diagnosis challenging and often delayed.4 There have been only about 500 diagnosed cases of CTX.5
References
- Nóbrega PR, Bernardes AM, Ribeiro RM, et al. Cerebrotendinous xanthomatosis: a practice review of pathophysiology, diagnosis, and treatment. Front Neurol. 2022;13:1049850. doi:10.3389/fneur.2022.1049850
- Freedman SF, Brennand C, Chiang J, et al. Prevalence of cerebrotendinous xanthomatosis among patients diagnosed with acquired juvenile-onset idiopathic bilateral cataracts. JAMA Ophthalmol. 2019;137(11):1312-1316. doi:10.1001/jamaophthalmol.2019.3639
- Köroğlu M, Karakaplan M, Gündüz E, et al. Cerebrotendinous xanthomatosis patients with late diagnosed in single orthopedic clinic: two novel variants in the CYP27A1 gene. Orphanet J Rare Dis. 2024;19(1):53-65. doi:10.1186/s13023-024-03082-4
- Stelten BML, Dotti MT, Verrips A, et al. Expert opinion on diagnosing, treating, and managing patients with cerebrotendinous xanthomatosis (CTX): a modified Delphi study. Orphanet J Rare Dis. 2021;16(1):1-18. doi:10.1186/s13023-021-01980-5
- Ejsmont-Sowała K, Książek T, Maciorowska-Rosłan K, et al. Case report: cerebrotendinous xanthomatosis treatment follow-up. Front Neurol. 2024;15:1409138. doi:10.3389/fneur.2024.1409138
Fragile X syndrome (FXS)
FXS is a neurological condition and is the leading inherited cause of intellectual disability.1
FXS is linked to a mutation in the FMR1 gene, which affects how brain cells communicate.2 This can lead to a range of symptoms, starting as early as 12 months of age.3 FXS occurs in approximately 1 in 4,000-7,000 males and 1 in 4,000-6,000 females in the United States.4-6
People living with FXS may experience developmental and behavioral challenges, including1-3:
- Learning disabilities
- Sensory processing challenges
- Anxiety
- ADHD
- Sleep disorders
- Seizures
There are no approved therapies specifically for FXS. However, there are behavioral interventions and medications prescribed off-label to treat symptoms of FXS. Research has shown that PDE4 inhibitors might be able to improve cognitive function, language, and daily activities in FXS patients.7
References
- Fragile X 101 | NFXF. National Fragile X Foundation. Accessed June 20, 2025. https://fragilex.org/understanding-fragile-x/fragile-x-101/
- Fragile X syndrome: MedlinePlus Genetics. Accessed June 20, 2025. https://medlineplus.gov/genetics/condition/fragile-x-syndrome/
- What is fragile X syndrome (FXS)? Cleveland Clinic. Accessed June 20, 2025. https://my.clevelandclinic.org/health/diseases/5476-fragile-x-syndrome
- Turner G, Webb T, Wake S, Robinson H. Prevalence of fragile X syndrome. Am J Med Genet. 1996;64(1):196-197. doi:10.1002/(SICI)1096-8628(19960712)64:1<196::AID-AJMG35>3.0.CO;2-G
- Hunter J, Rivero-Arias O, Angelov A, Kim E, Fotheringham I, Leal J. Epidemiology of fragile X syndrome: a systematic review and meta-analysis. Am J Med Genet A. 2014;164(7):1648-1658. doi:10.1002/ajmg.a.36511
- Ciaccio C, Fontana L, Milani D, Tabano S, Miozzo M, Esposito S. Fragile X syndrome: a review of clinical and molecular diagnoses. Ital J Pediatr. 2017;43(1):39. doi:10.1186/s13052-017-0355-y
- Berry-Kravis EM, Harnett MD, Reines SA, et al. Inhibition of phosphodiesterase-4D in adults with fragile X syndrome: a randomized, placebo-controlled, phase 2 clinical trial. Nat Med. 2021;27(5):862-870. doi:10.1038/s41591-021-01321-w
A pipeline builtfor impact
Our therapeutic focus areas are reflected in a pipeline designed with intention—advancing programs where scientific insight, patient need, and clinical clarity align.
From approved medicines to late-stage development, we are building a portfolio focused on delivering meaningful progress for patients living with rare diseases.
